COVID-19 vaccinations have begun in Japan. The most pressing issue in the smooth roll-out of the vaccination campaign is the creation of mechanisms for nationwide distribution of a vaccine requiring careful temperature control. Expectations are high that Panasonic's VIXELL cooling boxes are a key to overcoming this enormous challenge. We interviewed hospital staff administering the vaccines, VIXELL service providers, and VIXELL developers for answers on the innovative nature of the product and the contribution it is making in society.
Summary
- February 2021: COVID-19 vaccinations finally begin in Japan.
- U.S. Pfizer, the first vaccine approved for domestic use, must be transported at a range of "-75ºC±15ºC," "-25ºC to -15ºC," or "2ºC to 8ºC."
- This has put the spotlight on Panasonic-developed VIXELL cooling boxes.
- With the advantage of both strict temperature control and remote monitoring of conditions using IoT, here we introduce our product, incorporating the voices of hospital staff, service providers, and VIXELL developers.
COVID-19 vaccines--strict temperature control a must
Japan's COVID-19 vaccination drive began at last in February 2021, starting with health care practitioners. This foray into totally new ground is fostering unimaginable confusion, but one thing is certain--it has provided a ray of hope amid this COVID-19 crisis.
The complexity stems from the very delicate handling required by the vaccine. The first one approved for domestic use in Japan, Pfizer's vaccine must be transported and stored under strict temperature controls. The main vaccination centers in each prefecture (hospitals and clinics) all need deep freezers for storage. From there, small batches are distributed under refrigerated conditions of 2-8ºC to affiliated and satellite facilities (medical institutions, public venues, commercial facilities). Refrigerated delivery must be completed within 3 hours, and the defrosted vaccines can be stored at 2-8ºC for a maximum of 5 days.
In March 2021, new data showed that the vaccine could be stored for a maximum of 14 days between -25ºC and -15ºC meaning it could be kept in ordinary medical freezers. This simplified the process somewhat, but the vaccine rollout still requires extremely strict standards of temperature control.
Against that backdrop, Panasonic's announcement in January 2021 of VIXELL vacuum-insulated cooling boxes garnered much attention. That's because the revolutionary devices are the perfect answer for the demands of the COVID-19 vaccine, with their ability to maintain temperatures below -70ºC for 18 days when filled with dry ice.
Why did Panasonic focus on the distribution of medical and pharmaceutical products? We spoke to major pharmaceutical distributor Suzuken, developers of VIXELL, and staff at Matsushita Memorial Hospital in Moriguchi City, Osaka, where VIXELL boxes are used for in-hospital transport of COVID-19 vaccines. The interviews reveal the many possibilities for these highly functional cooling boxes.
Origins in strict temperature controls for specialty pharmaceuticals
A request from Suzuken led to the development of cooling boxes for pharmaceutical products in 2018. Suzuken distributes expensive specialty medical and pharmaceutical products, such as anti-cancer drugs, blood products, medicines for incurable diseases, and investigational drugs. When Suzuken started providing the Cubixx System for managing inventory, temperature and quality control in 2017, it represented an industry-first in medical and pharmaceutical distribution. A total traceability system, Cubixx uses contactless RFID tags attached to the products to track them as they move from the distributor to medical institutions where they are stored in dedicated storage cabinets run on IoT technologies. The system provides seamless data at every step in the process, from transport that conforms to Good Distribution Practices (GDP) to inventory management.
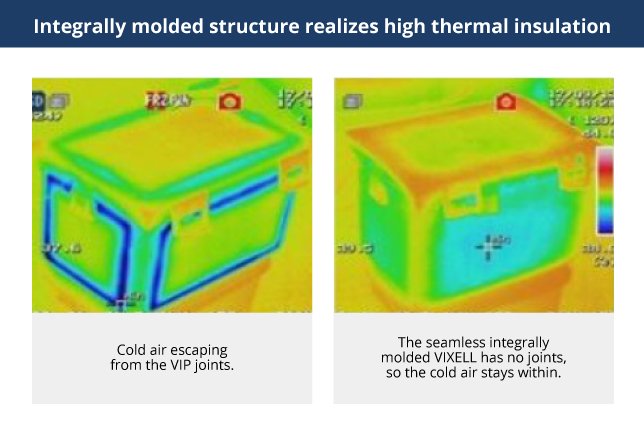
Instrumental in providing visibility for this pharmaceutical distribution system was the existence of cooling boxes that were easy-to-use and provided accurate tracking data. The original Cubixx system adopted cooling boxes with vacuum-insulated panels (VIP). Because they are constructed from individual insulation panels, cold air could easily escape from the connection seams. And because thicker panels are required for longer cooling times, the boxes became proportionately heavier. Finally, the aluminum foil surface covering inhibits the transmission of radio waves, preventing the temperature logging required in a traceability system.
That is when Suzuken and Panasonic started sharing ideas on designs for the ideal cooling box. Adoption of the seamless, integrally molded vacuum-insulated case (VIC), which Panasonic had been developing independently, simultaneously solved the issues of cold air leakage and box weight. One additional key point is the absence of metals like aluminum foil on the thermal storage surface and the adoption of resin instead, allowing for the transmission of radio waves.
The VIXELL design adopts a seamless integrally molded structure, simultaneously solving the issues of cold air leakage and weight.
"It was the combination of our pharmaceutical distribution expertise with Panasonic's technology for vacuum-insulated cases that enabled us to overcome key issues. The features of long-term, stable temperature retention and radio wave transparency made real-time temperature monitoring possible without ever opening the cooling box," says Seita Ando, Division Head for Medical and Home Care Solutions in the Healthcare Business Planning Department at Suzuken Co., Ltd.
Seita Ando
Medical and Home Care Solutions Division General Manager
Healthcare Business Planning Department
Suzuken Co., Ltd.
Those are not the only improvements offered by VIXELL. From 2021, a wireless vacuum sensor employing contactless power supply technology has been installed in the base of the vacuum-insulated cases of all models. The most important parameter in vacuum insulation is the vacuum degree, and this can now be checked simply by placing the box on a specialized inspection stand, providing peace of mind to users. Performance is further enhanced through links with IoT devices, such as internal temperature sensors and GPS, allowing users to monitor internal conditions without ever opening the box.
Conventional cooling boxes required thermal storage materials to be frozen at temperatures below zero prior to use. The cumbersome operation meant materials had to first be frozen before being raised to the 2-8ºC temperature range, thereby limiting applications. VIXELL adopts thermal storage material that solves this problem. A natural freezing process takes place when stored at 3ºC and no further adjustments to temperature are needed. Such improvements help to minimize temperature deviations and human error that can occur during transportation of specialty pharmaceuticals. "Being lighter than VIP cooling boxes is another advantage. Both men and women working at distribution sites have told us they feel more comfortable carrying these boxes," Mr. Ando explained.
The thermal storage material used in VIXELL means no further temperature adjustments are required, so long as it is stored at 3ºC. This minimizes temperature deviations and human error.
These virtues have drawn attention in the distribution of COVID-19 vaccines. At present, vaccines are transferred from central vaccination facilities to affiliate and satellite facilities using cooling bags distributed by the Ministry of Health, Labour and Welfare. These bags maintain a temperature below 8ºC for 12 hours when the outside temperature is 35ºC. With the planned acceleration in vaccinations, one process change is for medical and pharmaceutical distributors to spearhead the transport of vaccines. Mr. Ando says, "Many people are looking at VIXELL because of its ability to deliver goods at a constant temperature. This is necessary for vaccines, but it also opens up enormous possibility with regard to other pharmaceutical products, investigational drugs, and specimens that require strict temperature controls."
VIXELL's value and potential in practice
COVID-19 vaccinations for medical staff at Matsushita Memorial Hospital began in early April 2021. After arriving at the hospital, VIXELL was used to transfer vaccines within the hospital to the vaccination venue. "Innovation is essential for quality improvements in medical settings. We adopted VIXELL as part of that initiative," says Deputy Director Dr. Atsuhiro Hirata.
Dr. Atsuhiro Hirata
Deputy Director, Matsushita Memorial Hospital
Panasonic Health Insurance Organization
Head, TQM Center
Head, Innovation Promotion Division
Head, Medical Technology Division
Leader, Clinical Engineering Laboratory
In Japan, pharmaceutical companies ensure production and quality control of medical and pharmaceutical products based on the Good Manufacturing Practices (GMP) Ordinance. In 2018, "Good Distribution Practices (GDP) for Pharmaceutical Products" were developed, containing principles for high quality distribution and management of pharmaceutical products until they reach the relevant medical institution. The Cubixx System satisfies these guidelines.
As a pharmacist, Dr. Hirata also oversees the pharmaceutical department, and he points out that "We lacked the devices and infrastructure to properly maintain product quality in between arrival at the medical institution and delivery to patients. Temperature control is a key factor for hospital pharmaceutical departments handling a wide range of medical and pharmaceutical products. That's when the high-performance VIXELL arrived. Particularly in the case of the current Pfizer vaccine, there are time limits in addition to the need for temperature controls: 2 weeks between -25ºC and -15ºC, and 5 days if refrigerated at 2-8ºC. When removing products from regular cooling boxes, the simple act of opening and closing raises the temperature, but because VIXELL is highly insulated, the cooling agents rapidly reduce the temperature to the appropriate level after the lid is closed again. Previous cooling boxes were not equipped with this functionality, so I was very surprised."
Removing cooled vaccines prior to vaccination. Even if there are same-day cancellations, vaccines can continue to be stored and are not wasted when stored in VIXELL.
Each vial of the Pfizer vaccine is enough for five people, so the hospital had to arrange staff into groups of five to be vaccinated. This presented major scheduling difficulties, especially if people were unwell and needed to cancel on the day. "But using VIXELL as a temporary storage container, with its long-term cooling ability, we can continuously maintain strict temperature control and quality control. This enables us to save vaccines leftover due to same-day cancellations and safely using them at a later date. It's a great help that we can continue to store the vaccines in situations that would have required us to discard them in the past," added Dr. Hirata.
The high portability factor will be a great advantage as the vaccination campaign spreads to regular citizens. Dr. Hirata says, "There is no equipment to store medical and pharmaceutical products in aged-care facilities or gymnasiums. And there is concern around extreme heat in summer. When considering ways to store vaccines over many hours in those conditions, there is no question there is a need for cooling boxes like VIXELL." This also applies for transportation to remote islands, isolated areas and other geographical regions that are difficult to reach. That is because when transported under proper freezing conditions, even if vaccines are leftover, they can be taken back and used elsewhere.
Dr. Hirata went on to explain, "The same can be said in times of disaster. The fact is that many medical institutions do not have their own power generation source. I believe VIXELL is extremely helpful in maintaining a constant temperature without power. There are also applications in long-term storage of costly medical supplies and other precious medical resources, and for temporary storage in an operating theater--the possibilities are enormous."
A pediatrician administering vaccines shows great confidence in VIXELL: "Professionals in the pharmacy division dealing with these medical and pharmaceutical products implement strict temperature controls to make sure these vaccines don't go to waste, and this allows us to administer them with confidence." A registered nurse in the infection control division added: "I realize just how useful VIXELL is for the typically complicated issue of temperature control of vaccines. Thankfully, this enables us to focus all our efforts on giving vaccinations."
Growing need spawns VIXELL rental service
Shinya Kojima, VIXELL Business Project Leader in the Business Development Center of the Panasonic Appliances Company, has worked on vacuum-insulated panels for about 20 years since joining the company. The work originated in insulating panels for refrigerators, but the advancement came from the inclusion of Panasonic's existing technology for plasma display panels (PDP) in the independently developed vacuum-insulated case (VIC).
Shinya Kojima
VIXELL Business Project Leader
Business Development Center
Appliances Company
Panasonic Corporation
The integrally molded resin VIC produced after great difficulty was originally considered in product use cases, not as a component. But our surveys revealed an application in cooling boxes for medical and pharmaceutical products. Mr. Kojima reflects on the early days: "At our first meeting with Suzuken, there was nothing even resembling a box, but they put their trust in Panasonic and said, 'let's absolutely do this together.' Development took off immediately after that."
Panasonic had not anticipated working in the dry ice temperature range with the Cubixx system, but from around December 2020 when the reality of transporting COVID-19 vaccines set in, there was a sudden increase in inquiries on VIXELL's potential for stable temperature control at -75ºC±15ºC.
"Wishing to help resolve their issues, we conducted tests in the dry ice temperature range. The tests revealed that under constant conditions we could maintain a temperature of -70ºC for a maximum of 18 days, results we announced in a news release. There is nothing new in the B2B world about resolving customer issues, but we have only just started dealing with medical and pharmaceutical products, and the simple supply of VIXELL is not the end goal. We are being asked to resolve issues fast, but at the same time, new medicines are constantly being developed. This has shown us that unless we adopt the same speed, we cannot support medical innovation," Mr. Kojima noted.
In addition to temperature control, one of Panasonic's strengths is IoT. "The focus on incorporating IT not found in traditional cooling boxes was driven by our approach as an electrical appliance maker," adds Mr. Kojima. There has always been a need for real-time temperature surveillance during transport of medical and pharmaceutical products, so the announcement garnered a great response from companies dealing with the distribution of such products. This is perhaps also based on a major change in thinking in the pharmaceutical distribution industry based on the GDP guidelines.
Mr. Kojima went on to explain, "Initiatives are sprouting up around Japan for the distribution of pharmaceutical products under optimal conditions to help minimize losses. Still only guidelines in Japan, they have been enacted as laws in Europe and the U.S. I believe there will come a time for even stricter temperature management for pharmaceuticals than we're seeing now."
The current needs led to Panasonic and Suzuken Group joining forces to open a VIXELL rental service in April 2021. The business offers total fulfilment from rental bookings to pre-cooling, delivery to designated facility, and collection after use. Delivery here refers to the delivery of pre-cooled cooling boxes, not the transport of pharmaceuticals or vaccines, but there is no question that omission of the complicated process for pre-cooling and control is contributing to major efficiency gains. Mr. Kojima says the number of inquiries has prompted them to rapidly increase VIXELL production.
Demand for VIXELL is increasing: the IT-enabled cooling boxes reduce barriers to pharmaceutical management.
Like Dr. Hirata, Mr. Kojima expects to see growing application of VIXELL as a temporary storage mechanism for COVID-19 vaccines in remote islands and isolated regions. In addition, the COVID-19 crisis has caused patients to limit their hospital visits, so there is growing demand for the refrigerated transport of medicines prescribed through online consultations. Suzuken and pharmaceutical companies have already started using VIXELL in situations such as this in a service known as Direct to Patient (DTP) transport. With its sights also on international transport of refrigerated pharmaceuticals and experimental drugs from overseas to Japan, Panasonic added a larger volume box to its lineup in 2020.
These advancements are making significant contributions to the medical industry. Mr. Ando expresses a desire to "take the know-how cultivated at Suzuken on transport using VIXELL and share it with the medical industry to develop the traceability systems into platforms of medical infrastructure." Mr. Kojima also speaks of future prospects: "As with DTP, we hope to evolve the product for applications in home-visit medical care and virtual clinical trials, based on feedback from various fields."
Dr. Hirata adds his hopes for the future: "Wouldn't it be ideal to use VIXELL at every step in the process from pharmaceutical company to distributor, then onto medical facility and within the hospital too? As someone on the medical frontlines, ultimately my goal is to maintain the quality of pharmaceuticals until the instant before they enter a patient's body. From that perspective, I believe the applications for VIXELL are endless."
# # #
Disclaimer:
We would like to note that Panasonic Newsroom is not a place to address personal Customer Service issues. Even though this is not the forum, Panasonic is always eager to resolve your concerns. Our local customer services contacts can be found at Global Support or you can see our list of Social Media Accounts to find the right channel for your queries and concerns.
Related News
Related Videos
VIXELL™ - A Vacuum-insulated Cooling Box for -70ºC Storage